[ad_1]
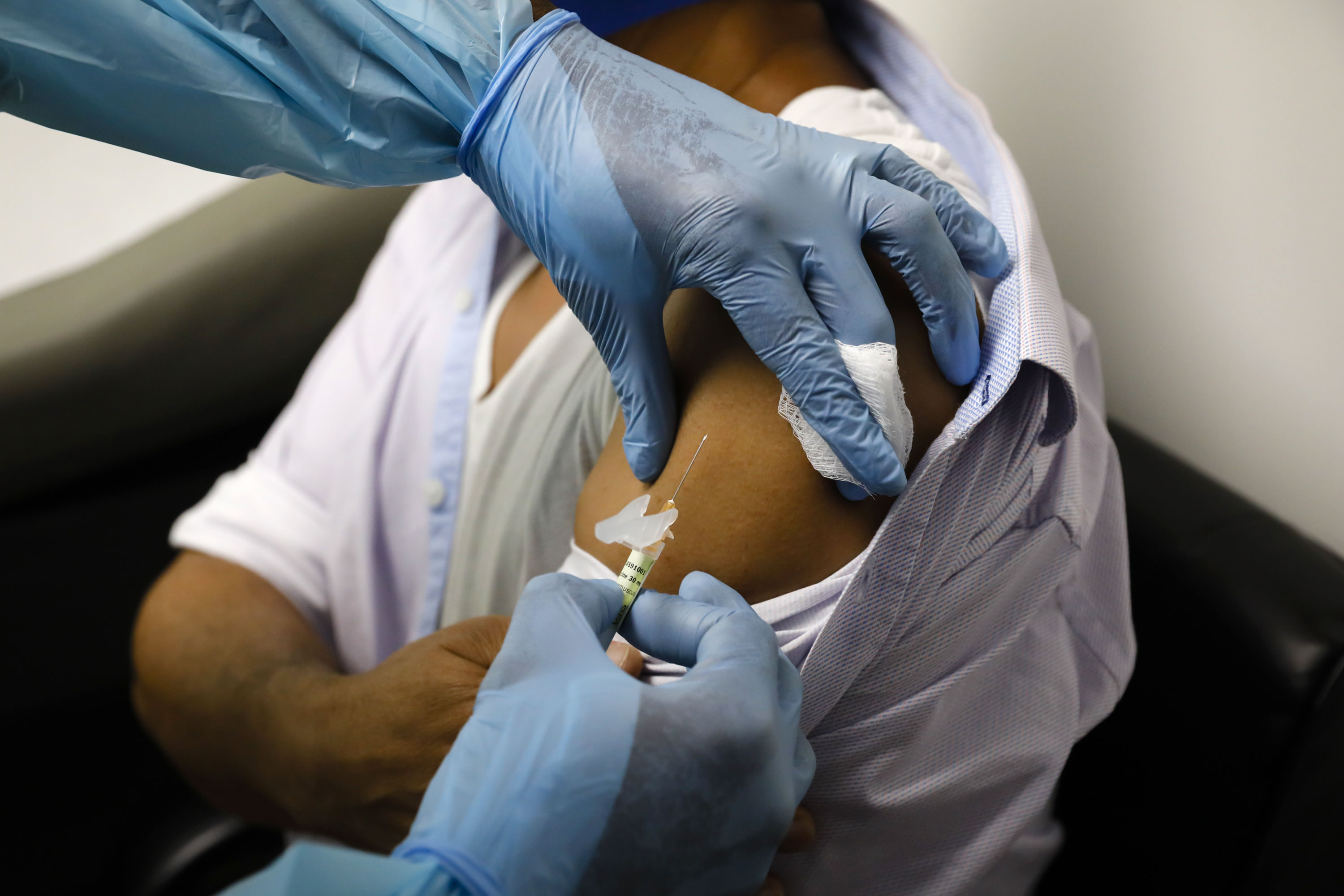
(CNN) – The UK will receive its first batch of Pfizer-BioNTech’s COVID-19 vaccine in the next few hours, England’s deputy medical director said Thursday.
“We hope to achieve [la vacuna Pfizer] very early in the UK, and I mean hours, not days, ”Jonathan Van-Tam told the BBC’s Five Live radio show.
WATCH: “70% need to be vaccinated to talk about the end of the pandemic,” according to a specialist
On Wednesday, the UK Department of Health said up to 800,000 doses of the vaccine would be available next week, enough to inoculate 400,000 people.
Van-Tam also said he believes US and European regulators will approve Pfizer’s COVID-19 vaccine in the next “days or weeks,” during a separate interview with BBC Breakfast.
“I don’t think other regulators, like those in the US, will take this vaccine long. I think this will be resolved in the next few days, ”Van-Tam said.
Fauci says the FDA is more careful
His comments come after Dr Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said British health regulators who licensed Pfizer’s covid-19 vaccine on Wednesday did not review the study data with the same attention. as the US Food and Drug Administration (FDA) is doing in its review.
“The way the FDA is, our FDA is doing it, it’s the right way,” Fauci said in an interview with Fox News. “We have really looked at the data very carefully to assure the American public that this is a safe and effective vaccine,” he said.
These groups will have priority to receive the UK coronavirus vaccine
Van-Tam explained that the UK vaccination program will prioritize the elderly in nursing homes and their workers, hence “all people aged 80 and over” and “those with unsafe conditions”.
“More vaccines are on the way, we have invested in seven more and they should start arriving in the spring,” Van-Tam added.
The chief epidemiologist also told the BBC that pregnant women should not be vaccinated at this time: ‘We don’t have the data at this time. There might not be any problem, but safety comes first. “
As for the children, Van-Tam added that they are not a priority at the moment, but that the producers “are now thinking about testing children”.
The deputy medical director also urged priority groups to accept the vaccination request, saying that the success of the vaccination program and the subsequent lifting of restrictions depend on public adherence to the plan.
WATCH: What could be the main obstacles to vaccine distribution in Argentina?
“It is not my job to give them a magic number because it all depends on how quickly the vaccine is released, if the people called to receive the vaccine show up. If it turns out to be 10%, this vaccine, even if perfect, will have no effect on public health at the population level, “he added.
As for the duration of immunity provided by the Pfizer vaccine, Van-Tam said there is still no certainty “how long it will last”.
“We have some evidence that these vaccines will stimulate T cells and antibodies, which could last a long time. But we don’t know how long it will last. I’m sure it will take several months at a minimum,” he added.
Van-Tam said there are still some “challenges in terms of storage conditions” regarding the Pfizer vaccine, which must be stored at minus 70 degrees, but there are also a limited number of times the vaccine can. ” transit through the ambient air temperature “.
He added that he hopes the AstraZeneca-Oxford vaccine, which “ should be much easier and more deployable in more NHS settings. [Servicio Nacional de Salud]”, It could be” as easy as a flu shot “to manage.
Van-Tam also added that he is “confident” that the AstraZeneca-Oxford vaccine will be ready by Christmas.
Source link