[ad_1]
The vision of the future of miniaturization has produced a series of synthetic molecular motors that are driven by a variety of energy sources and can perform various movements. A research team from the Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU) has now managed to control a catalysis reaction using a controlled light engine. This brings us one step closer to realizing the vision of a nano factory where combinations of various machines work together, as in the case of biological cells. The results were published in Journal of the American Chemical Society.
The laws of mechanics cannot always be applied
By definition, an engine converts energy into a specific type of kinetic energy. At the molecular level, for example, the myosin protein can produce muscle contractions using chemical energy. Such nanomachines can now be synthetically produced. However, the molecules used are much smaller than proteins and significantly less complex.
“The laws of mechanical physics cannot simply be applied at the molecular level,” says Prof. Dr. Henry Dube, Chair of Organic Chemistry I at FAU. Inertia, for example, doesn’t exist at this level, he explains. Triggered by Brownian motion, the particles are constantly in motion. “Activating a rotary motor is not enough, you need to incorporate some type of ratchet mechanism that prevents it from rotating backwards,” he explains.
In 2015, during the LMU in Munich, Prof. Dube and his team developed a particularly fast molecular motor driven by visible light. In 2018 they developed the first molecular motor that is driven solely by light and operates regardless of ambient temperature. A year later, they developed a variant capable not only of rotation but also of performing a figure eight movement. All engines are based on the hemithioindigo molecule, an asymmetrical variant of the natural indigo dye in which a sulfur atom takes the place of the nitrogen atom. One part of the molecule rotates in stages in the opposite direction to the other part of the molecule. Energy-driven passages are activated by visible light and modify molecules to block reverse reactions.
Standard catalysts in use
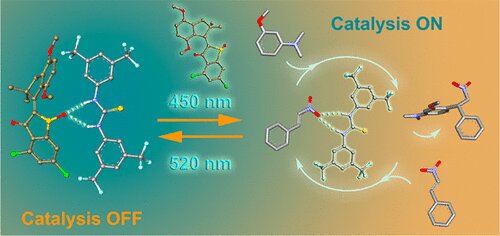
After arriving at FAU, Henry Dube first used the rotary motor developed in 2015 to control a separate chemical process. It moves in four phases around the carbonaceous double bond of hemi-indigo. Two of the four steps triggered by a photo reaction can be used to control a catalysis reaction. “Green light generates a molecular structure that binds a catalyst to the hemi-indigo and blue light releases the catalyst,” explains the chemist.
A standard catalyst that does not have metal atoms is used. Using electrostatic forces, the catalyst engages via a hydrogen bond on an oxygen atom in the ‘motor molecule. “In principle, all catalysts that use a hydrogen bond could be used.” The great advantage of hemithioindigo is that its innate structure has a binding mechanism for catalysts, “explains Prof. Dube. If not, it should be added using chemical synthesis.
The rotation of the hemi-indigo motor is controlled by visible light. At the same time, the system allows for the targeted release and binding of a catalyst that accelerates or decelerates the desired chemical reactions. “This project is an important step towards integrating molecular motors into chemical processes in a simple way and in various ways,” says prof. Dube. “This will allow us to synthesize complex drugs at a high level of precision using molecular machines as a production line in the future.”
Rotation on a figure-of-eight path
Kerstin Grill et al, Relè Supramolecular Relay-Control of Organocatalysis with a Hemithioindigo-Based Molecular Motor, Journal of the American Chemical Society (2020). DOI: 10.1021 / jacs.0c09519
Provided by the University of Erlangen-Nuremberg
Quote: Control catalysis of controlled light nanomachines (2020, November 23) retrieved November 23, 2020 from https://phys.org/news/2020-11-light-controlled-nanomachine-catalysis.html
This document is subject to copyright. Aside from any conduct that is correct for private study or research purposes, no part may be reproduced without written permission. The content is provided for informational purposes only.
[ad_2]
Source link